
Inform patients of the signs and symptoms of anaphylaxis, and instruct them to seek immediate medical care should signs or symptoms occur. Observe patients closely for an appropriate period of time after administration of XOLAIR, taking into account the time to onset of anaphylaxis seen in premarketing clinical trials and postmarketing spontaneous reports. Initiate XOLAIR only in a healthcare setting equipped to manage anaphylaxis which can be life-threatening. Approximately 60% to 70% of anaphylaxis cases have been reported to occur within the first three doses of XOLAIR, with additional cases occurring sporadically beyond the third dose. In postmarketing spontaneous reports, the frequency of anaphylaxis attributed to XOLAIR use was estimated to be at least 0.2% of patients based on an estimated exposure of about 57,300 patients from June 2003 through December 2006. The time to onset of anaphylaxis was 90 minutes after administration in two patients and 2 hours after administration in one patient.Ī case-control study showed that, among XOLAIR users, patients with a history of anaphylaxis to foods, medications, or other causes were at increased risk of anaphylaxis associated with XOLAIR, compared to those with no prior history of anaphylaxis. Anaphylaxis occurred with the first dose of XOLAIR in two patients and with the fourth dose in one patient. In premarketing clinical trials in patients with asthma, anaphylaxis was reported in 3 of 3507 (0.1%) patients. WARNINGS AND PRECAUTIONSĪnaphylaxis has been reported to occur after administration of XOLAIR in premarketing clinical trials and in postmarketing spontaneous reports. XOLAIR is contraindicated in patients with a severe hypersensitivity reaction to XOLAIR or to any ingredient of XOLAIR. *Programs have specific eligibility criteria. 
If you think your patient qualifies for XOLAIR Access Solutions, submit the completed Prescriber Service Form and Respiratory Patient Consent Form to Genentech Access Solutions. With My Patient Solutions, you can: enroll patients online* view and track your patient cases view Benefits Investigation (BI) reports get case status alerts and request benefits reverification for multiple patients at once. 
My Patient Solutions ®, a service provided to give you the flexibility to submit and check the status of your XOLAIR Access Solutions patient cases when it's convenient for you.The XOLAIR Starter Program, which helps your patients get access to XOLAIR while you're waiting for insurance authorizations.Product Distribution-learn about specialty pharmacies in your area. 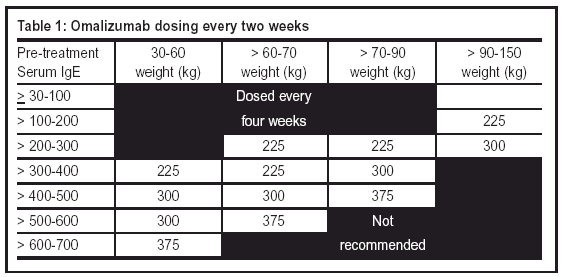
On the Access Solutions website, you can also find information about the following topics:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
